Projects
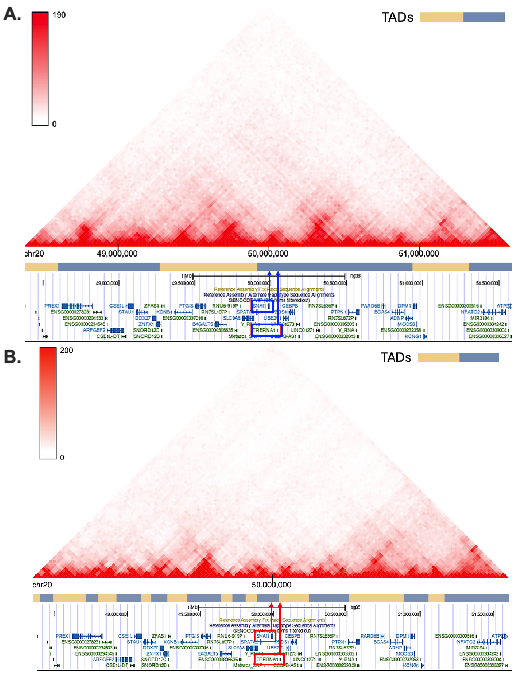
Using DPP4 inhibitors as a probe, we elucidate a novel chromatin remodeling pathway in the lens driving EMT during posterior capsule opacification, then exploited it to design a CRISPR-Cas9 gene therapy that replicates the topological effect without the drug's cytotoxicity.
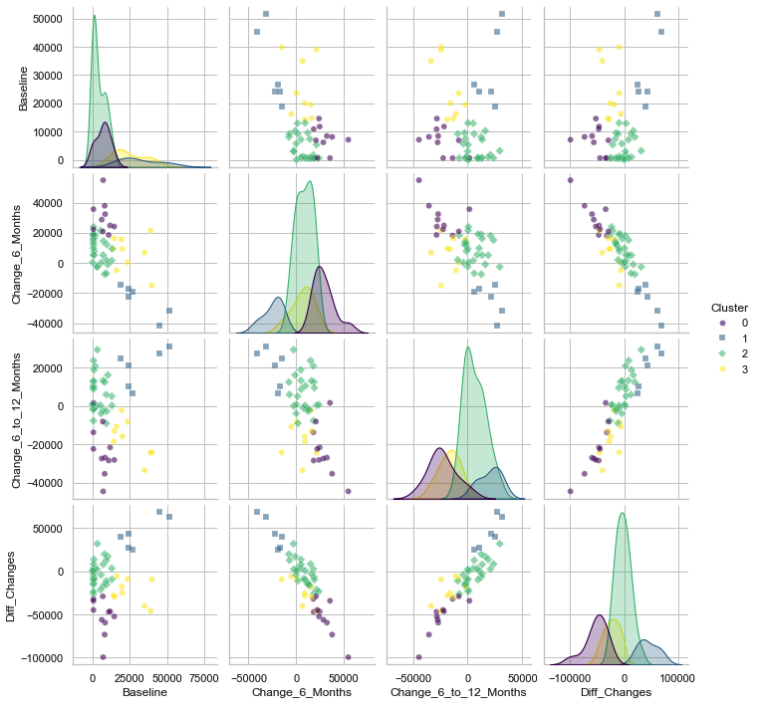
Created an easy-to-use pipeline via conditional VAEs to learn latent features present within trajectories of brain-derived neurotrophic factor measurements in pediatric patients. Determined that trajectories were able to correlate to behavioral outcomes post-injury. Pipeline used in multiple NIH-funded studies at UPMC Children's Hospital.
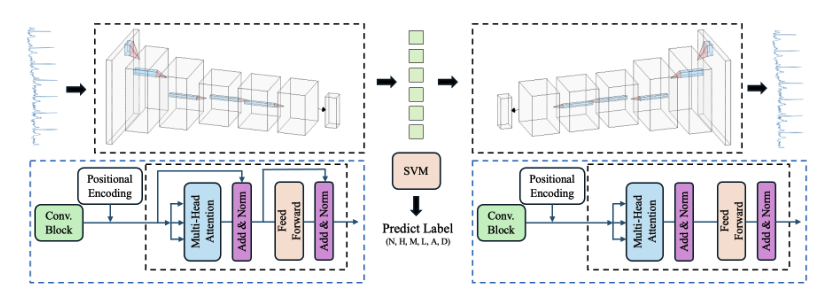
A screening pipeline that uses an Apple Watch as a 12-lead ECG; We developed a regression model to upscale 4-lead data to 12 leads, and a transformer auto-encoder to classify beats at 95.3% sensitivity and 99.1% specificity, outperforming physicians and SOTA algos on the same dataset.